The commonly used 4H-SiC and 6H-SiC space groups are both P63mc, and the point group is 6mm. 6mm belongs to one of 10 polar point groups (1, 2, 3, 4, 6; m, 3m; mm2, 4mm, 6mm), so 4H-SiC and 6H-SiC are polar crystals. The polar crystal refers that at least one direction in a crystal has different properties from the opposite direction, which can be electrical properties (thermoelectric properties, ferroelectric properties), growth properties, etc. The SiC crystal is a typical polar crystal. The silicon carbide polarity will be explained in the following part.
According to Newman’s law, the symmetry of physical properties is higher than the symmetry of crystals. This asymmetrical polarity is due to the fact that the microscopic electric dipoles inside the crystal have the same orientation or tend to be the same in this direction, and they do not cancel each other out. This direction is called the polar axis, and the polar axis corresponding to the crystal plane is the polar plane.
The silicon carbide polarity affects the growth. Polar crystals have different polar growth rates. Generally, the growth rate of the positive side is higher than that of the negative side. A crystal can have multiple polar directions and multiple sets of polar.
And the growth affects performance. SiC devices generally use a silicon surface for epitaxial growth, and the bottom surface of the substrate is a carbon polar surface. In fact, the epitaxy of different materials with different polar planes will show different properties.
1. Calculation of the Silicon Carbide Polarity
If the crystal orientation is determined according to the cif file, the atomic number is 14/6 through R0=1.883; bringing in the atomic coordinates, the [SiC4] tetrahedral dipole moment is mainly along the -c axis. The direction of the dipole moment is that the negative pole points to the positive pole, which can correspond to the Si surface as (000-1) and the C surface as (0001).
Definition: The orientation <0001> of a chemical bond of Si atoms in a silicon tetrahedron corresponds to the Si plane, and the orientation <000-1> of a chemical bond of C atoms in a carbon tetrahedron corresponds to the crystal plane C.
![]()
The Si-C bond polarity in that direction is separated from the vertical plane in the c direction, the Si surface is the silicon surface, and the C surface is the carbon surface.
![]()
The Si surface is (0001), and the C surface is (000-1).
Therefore, the crystal plane diagram is as follows:
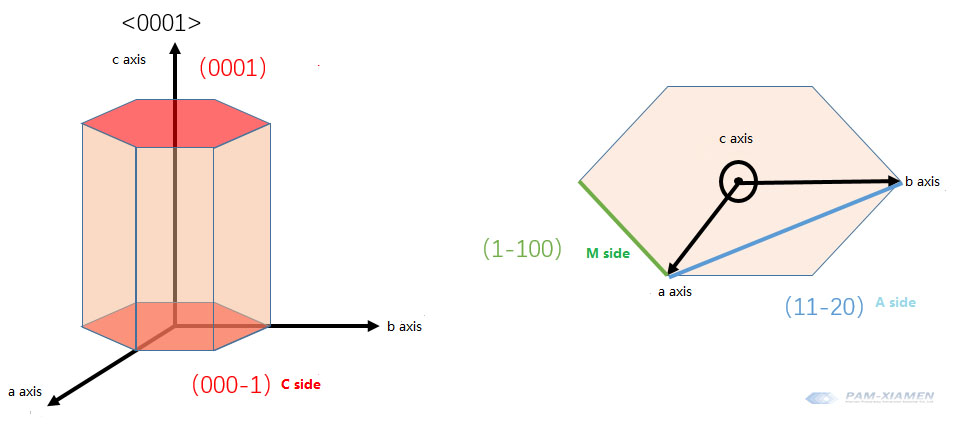
The standard wafer is that (0001) with a few degrees off angle points to (11-20). The off angle is used for step flow growth of silicon carbide. If you grow wafers, like GaN wafer, you can have no deflection angle.
The orientation before cutting is to determine the three directions of <0001><11-20><1-100>.
2. The Determination for SiC Polar
Actually, the determination for the polarity of silicon carbide is to use various instruments to find the difference between the two surfaces. Corrosion method to determine the SiC polarity structure has related introductions.
2.1 Why Choose the Erosion Method for Determination of the Silicon Carbide Polarity?
Firstly, the X-ray abnormal absorption method cannot be used to judge. The X-ray anomalous absorption method is feasible for InSb and GaAs. But due to the low atomic numbers of carbon and silicon, it is difficult to obtain the wavelength corresponding to the absorption limit, so it is difficult to use this method to determine the silicon carbide polarity.
Secondly, the crystal growth shape cannot be used to judge. Sometimes the whole furnace SiC produces a wafer that one side is a large and flat, and another side is curved wedge-shaped. The growth shape is mainly affected by external growth conditions, and the curved surface is mainly produced by thermal erosion. The orientation of the curved surface and the flat surface is very regular, the flat surface faces the low temperature zone, and the curved surface faces the high temperature zone.
2.2 The Erosion Method for Determination of the SiC Polarity
According to the fact that diamond does not react with molten alkali, but silicon can react with molten alkali, it can be considered that the less corroded side is the carbon side, and the corroded side is the silicon side. Molten potassium hydroxide is etched at a temperature of 600-800°C for 10 minutes and then taken out for washing. Erosion pits may appear on both sides and the shape is generally hexagonal.
However, the ratio of the depth and linearity (diagonal length) on both sides is different: the larger is the silicon surface, and the smaller is the carbon surface. The disadvantage of this method is that it causes at least 40um damage to the two surfaces of the silicon carbide wafer. Only after polishing, the etched wafer can be reused. The molten KOH up to 500 degrees Celsius has a large damage rate to the wafer, and molten alkali in the open state is potentially dangerous to operators.
Corrosion in the polishing process has also been proposed for measurement. A method for identifying the silicon-carbon surface of a silicon carbide wafer refers to two surfaces that are also polished: if the displayed roughness value is between 0.10 and 0.50 nm, the tested surface is a silicon surface; if the roughness is between 0.80~3.00nm, the tested surface is a carbon surface. In other words, the carbon surface has a higher roughness, and more and deeper scratches. However, the comparative study of CMP polishing of SiC (0001) surface and (000-1) surface pointed out that after polishing, the roughness of silicon surface is greater than that of carbon surface. The surface roughness of the silicon surface is between 0.2 and 2.0 nm; the roughness of the carbon surface is between 0.1 and 0.3 nm, and the tested surface is a carbon surface.
The interesting thing is that the polishing rate can be used to identify the polarity of silicon carbide, which is also conflicting in the literature. The comparative study on 6H-SiC (0001) surface and (000-1) surface CMP polishing pointed out that using the modified silica sol polishing solution with pH values of 10.38 and 1.11 for polishing, the removal rate of carbon surface is higher than that of silicon surface; the removal rate of acid polishing liquid is fast. The research concluded that the removal rate of silicon surface material is the highest, the m-side and a-side surface are lower than the silicon surface, and the carbon surface material removal rate is the lowest and almost zero.
In fact, the polarity may have exhibit ferroelectric and thermoelectric properties, which can also be used.
3. Marking of the Silicon Carbide Polarity
The growth process of the silicon carbide polarity can be maintained. So marking is very important and necessary. At present, the commonly used method for distinguishing the silicon carbide surface is to orient the ingot twice after spheronization of the silicon carbide single crystal, and process two straight surfaces of different sizes, called the primary and secondary reference planes. After slicing, determine the silicon carbide polarity by the sort of the primary and secondary reference planes.
However, this method has two shortcomings. Firstly, to distinguish between the primary and secondary reference planes, the length of the primary reference plane is longer. The primary reference plane of a 4-inch silicon carbide single crystal is as long as 32.5 mm, and that of the 6-inch silicon carbide single crystal is 47.5 mm long, which wastes the cost, reduces the usable area of the substrate, and damages the crystal ingot. Secondly, when a wafer with a long reference edge is used as a material crystal, it has a great influence on the quality of the grown single crystal. It is easy to cause defects to gather near the main and auxiliary reference edges. Moreover, once the crystal breaks, it is difficult to distinguish the polarity of the chip by this method.
Some improvements are unequal marking edges. A method for identifying the polarity of c-si of a silicon carbide wafer adopts an asymmetric V-shaped groove to calibrate the positive and negative polarities. Or add laser marking to the auxiliary positioning side.
For more information, please contact us email at victorchan@powerwaywafer.com and powerwaymaterial@gmail.com.

